 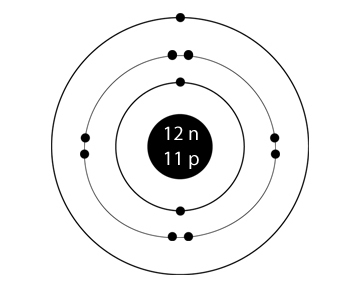
So, for counting the number of lectrons in the first shell we again apply the rule as:. How many electrons should be added in the shell is given by #2n^2# rule where n refers to the number of shell. 
How do we know how many eletrons should be added to the first shell? STRUCTURE FOR SODIUMĮlectronic configuration:- K - 2, L-8, M-1 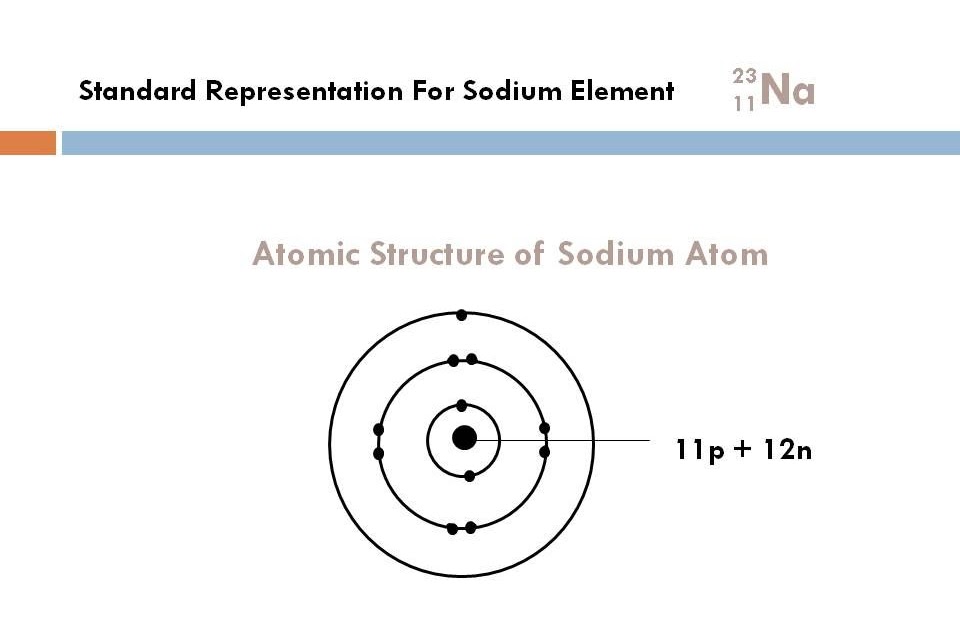
The number of elecrons each shell of an element can old is given by #2n^2# rule given by Bohr where n refers to the number of shell. We always do electronic configuration for the number of electrons present in the atom of the element. Do the electronic configuration for the atom of the element. Therefore, now you know the number of electrons too. As in every stable atom the number of protons is equal to the number of electrons. Once you know its atomic number you know the number of protons and electrons in it. Always write the atomic number of the element first. Atomic number of any element refers to the number of electrons in an atom of that element they are having. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium.Ītomic number of sodium is 11. That is, in this case, the valency of the sodium ion is +1. The electron configuration shows that the sodium atom has acquired the electron configuration of neon. The electron configuration shows that sodium ion (Na +) have only two shells and the last shell has eight electrons. It may also be produced by the thermal decomposition of sodium azide.We draw atomic structures for any element with the help of atomic number they have. The electron configuration of sodium ion (Na +) is 1s 2 2s 2 2p 6. Pure sodium may be obtained by electrolysis of molten sodium chloride. Sodium was first commercially produced by thermal reduction of sodium carbonate with carbon at 1,100 degrees Celcius, in the Deville process.The most common sodium mineral is halite or sodium chloride salt. Although it's too reactive to occur in pure elemental form, it is found in many minerals, including halite, cryolite, soda niter, zeolite, amphibole, and sodalite.It is the sixth most abundant element on Earth, comprising about 2.6% of the earth's crust. It's found in the sun and many other stars. The symbol for sodium is Na, which comes from the Latin natrium or Arabic natrun or a similar-sounding Egyptian word, all referring to soda or sodium carbonate.There is only one stable isotope of sodium: 23Na.Sodium and its compounds are used for food preservation, for cooling nuclear reactors, in sodium vapor lamps, for purifying and refining other elements and compounds, and as a desiccant.In humans, sodium is important for maintaining fluid balance in the cells and throughout the body, while the electric potential maintained by sodium ions is critical for nerve function. This process is known as electrolysis and using it Davy went on to isolate elemental potassium, calcium, magnesium and barium by a very similar method. Sodium is an essential element in animal nutrition. It was this terminology that inspired Sir Humphrey Davy to call the element sodium when he first isolated it by passing an electric current through caustic soda, or sodium hydroxide, in 1807.At room temperature, sodium metal is soft enough that you can cut it with a butter knife.The pure metal is stored in oil or kerosene because it spontaneously ignites in water. Sodium is a silvery-white metal belonging to Group 1 of the Periodic Table, which is the alkali metals group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
